According to the National Cancer Institute, over 2 million Americans were diagnosed with cancer in 2025. The average age of someone who receives a cancer diagnosis is 65-74 years. But young people can develop cancer too, and their cancers can be quite distinct from adult cancers. Pediatric cancer patients would benefit from personalized approaches to their diseases to improve treatment outcomes.
New research by Dr. Jason Berman, CEO and Scientific Director of the CHEO Research Institute in Ottawa, Canada and Professor of Pediatrics and Cellular and Molecular Medicine at the University of Ottawa, and Dr. Jamie Fletcher, Associate Director of Laboratory Research and Principal Scientist at Children’s Cancer Institute in Sydney, Australia and Associate Professor in the Faculty of Medicine at UNSW Sydney, and their Canadian and Australian colleagues shows that zebrafish models of pediatric cancer are an additional personalized medicine tool that can be incorporated into pediatric cancer treatment.
Precision Medicine for Pediatric Cancers
The American Childhood Cancer Organization estimates that over 15,000 children and adolescents are diagnosed with cancer each year. Pediatric cancer1Pediatric cancer: A rare genetic disease that occurs in children and adolescents. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/pediatric-cancer is a group of rare genetic diseases where certain genetic mutations2Mutation: A change in the genetic sequence of an organism; most mutations don’t have consequences but some can affect human health. https://www.genome.gov/genetics-glossary/Mutation lead to abnormal cell growth. Although all pediatric cancers are rare, the most frequent types of pediatric cancers diagnosed include tumors of the brain and spinal cord, leukemia3Leukemia: Type of cancer that affects the red blood cells. https://www.cancer.org/cancer/types/leukemia-in-children.html, and neuroblastoma4Neuroblastoma: Type of cancer that develops from immature nerve cells. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/neuroblastoma. Less common types of pediatric cancer include Ewing sarcoma5Ewing sarcoma: Type of cancer that affects the bones and connective tissues. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/ewing-sarcoma, osteosarcoma6Osteosarcoma: Type of aggressive bone cancer that usually affects the large bones of the arms or legs. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/osteosarcoma, gastrointestinal stromal tumors7Gastrointestinal stromal tumor: Type of cancer that begins in the lining of the gastrointestinal tract. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/gastrointestinal-stromal-tumor, and anaplastic large cell lymphoma8Anaplastic large cell lymphoma: Aggressive type of cancer that affects specialized cells of the immune system. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/alcl.
The mainstay of pediatric cancer treatment is chemotherapy9Chemotherapy: The use of drugs to kill cancer cells. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/chemotherapy, which is also a common treatment option for adult cancers. Chemotherapy is the use of drugs given by mouth, injection, or infusion to kill cancer cells. While chemotherapy in children and adolescents can work well, it also comes at a high cost. “Using chemotherapy to treat pediatric cancer is like using a cannonball to kill a fly,” explained Dr. Berman. “You can effectively kill the cancer, but there is significant collateral damage.”
In the case of chemotherapy, side effects from treatment can last a lifetime or lead to other health issues years down the road. An estimated two-thirds of pediatric cancer survivors will develop at least one chronic health condition because of their cancer or cancer treatment. This high cost may be worthwhile when the chemotherapy works. Unfortunately, even when chemotherapy treatments initially kill the cancer, the disease can sometimes come back. When this happens, there are very limited treatment options.
Given the challenges of treating patients with pediatric cancer, scientists have moved towards a more personalized treatment approach called precision medicine10Precision medicine: A personalized approach to health care that uses information about an individual’s genes and environment to diagnose and treat disease. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/precision-medicine. Precision medicine allows clinicians and researchers to work together to identify specific treatment targets based on the genetic mutations of an individual patient’s cancer.
Collaboration Across Continents
A unique feature of pediatric cancer research is the way that doctors who treat pediatric cancers, called pediatric oncologists11Oncologist: A doctor who specializes in diagnosing and treating cancer. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/oncologist, and researchers who study pediatric cancers work together. Since pediatric cancers are so rare, even national pediatric cancer centers receive relatively few patients with each kind of cancer. As a result, national centers often collaborate across international borders to test new treatment options in clinical trials12Clinical trial: A research study with human participants designed to test the effectiveness of specific health interventions. https://grants.nih.gov/policy-and-compliance/policy-topics/clinical-trials/definition. These centers are familiar with their counterparts in other countries and are connected by their shared mission of treating patients with pediatric cancer.
The commitment to international collaboration among the pediatric cancer research community brought Dr. Berman and the Canadian national precision medicine program, PRecision Oncology For Young peopLE (PROFYLE), together with their Australian counterparts, including Dr. Jamie Fletcher and the Australian ZERO Childhood Cancer precision medicine program.
Honing Precision Medicine Techniques for Pediatric Cancers
Dr. Jamie Fletcher is a pediatric cancer researcher who specializes in precision medicine techniques including developing cellular and animal models of pediatric cancer and using these models to test different treatment options. He is also the co-scientific lead of the preclinical13Preclinical study: Research conducted using animal models to test the safety and effectiveness of specific health interventions before they are tested in clinical trials. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/preclinical-study drug testing team at the ZERO Childhood Cancer program.
When a new patient enrolls in the ZERO program, Dr. Fletcher and his team receive a sample of the patient’s tumor tissue for testing. They use state-of-the-art laboratory equipment to analyze the samples and identify genetic mutations that might be the underlying cause of the cancer. In addition, Dr. Fletcher and his team use these samples to grow tumor cells in the laboratory. The researchers have a library of treatment options approved for pediatric use that are known to target tumors with specific genetic mutations, and they test possible treatment options on the tumor cells to see how they respond. Using these techniques, Dr. Fletcher and his team can identify recommended treatment options based on the specific genetic profile of a patient’s tumor in 60-70% of cases.
“It’s great that we are able to guide treatment recommendations for so many pediatric patients across Australia,” remarked Dr. Fletcher, “but that still leaves about one-third of patients who we are not able to help.” For these patients, Dr. Fletcher and his colleagues have become experts in the use of specialized mouse models called patient-derived xenograft14Patient-derived xenograft: Tumor tissue taken from a human patient and transplanted into an animal model for research. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/patient-derived-xenograft (PDX) models. Xenograft15Xenograft: The transplantation of tissue, such as tumor cells, from one species to another. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/xenograft refers to the transplantation of tissue from one species to another, in this case from human to mouse. Researchers take a sample of tumor cells from a patient and grow them in a mouse model rather than in a laboratory dish. They then test how the tumors in the mouse PDX models respond to different treatment options.
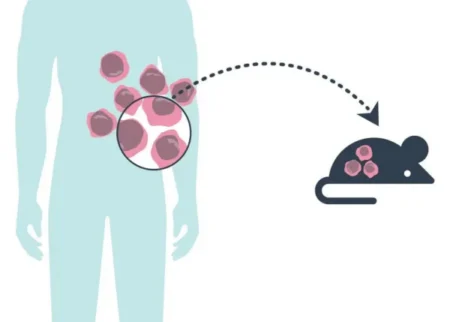
To make a patient-derived xenograft mouse model, researchers take tumor tissue samples from a human patient and graft them into a mouse model.
[Source: https://www.cancer.gov/news-events/cancer-currents-blog/2021/cancer-pdx-mouse-models-retain-genetics-human-tumor]
In the ZERO program, Dr. Fletcher and his team attempt to develop mouse PDX models for all enrolled patients. When the results from testing in cell lines and PDX models agree, it can give clinicians even higher confidence that the recommended treatment will be effective for an individual patient. For patients whose tumors don’t have any identifiable genetic mutations causing disease, the PDX models can provide valuable information.
However, there are still many barriers that need to be overcome to make PDX modeling even more relevant for patients. The researchers are only able to develop mouse PDX models for about half of patients. This is because not all attempts at grafting the tumor cells are successful, depending on the specific type of cancer cells and the amount of cells available.
In addition, it can take many months before a mouse PDX model is mature for drug testing, and patients often don’t have the time to wait on a result before starting a new treatment. Treatments used while waiting for the model can change the genetics of the tumor such that the model is no longer as relevant. “Mouse PDX models are really wonderful tools,” added Dr. Fletcher, “but it’s really difficult to turn these models into treatment recommendations for patients within the time frame that patients need.” Zebrafish are an attractive alternative to mouse models because they can be developed in a much shorter time frame.
Zebrafish Models of Pediatric Cancer
On the opposite side of the globe from Dr. Fletcher and colleagues in Australia, Dr. Berman and his team in Canada also work to address the challenges of precision medicine in pediatric oncology. Dr. Berman is a pediatric oncologist specializing in leukemia and a pediatric cancer researcher whose research focuses on the use of zebrafish models for precision medicine.

Zebrafish are a common animal model of disease used since the early 2000s.
[Source: https://commons.wikimedia.org/wiki/File:Zebrafisch.jpg]
Dr. Berman began working with zebrafish in the early 2000s when research on zebrafish as models of human disease was just beginning. In his research career, Dr. Berman has used zebrafish to develop precision medicine approaches for pediatric cancers in numerous ways, including the development of PDX models of pediatric cancers in zebrafish.
In the zebrafish PDX model Dr. Berman developed, tumor cells are injected into the zebrafish at two days of age. At Day 3, treatments are added into the water and absorbed by the zebrafish. Over the next 4-7 days, the researchers observe what happens and analyze the results.
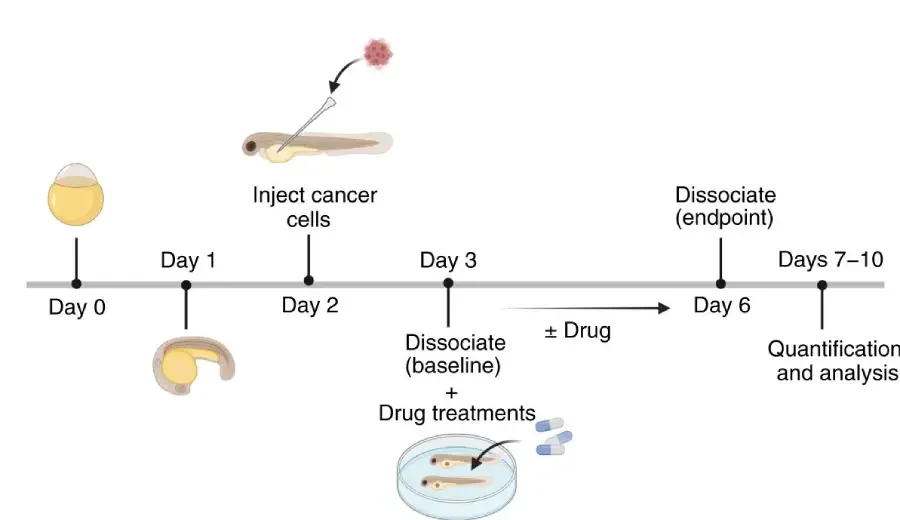
Process for testing drug treatments using a zebrafish PDX model.
[Source: Azzam et al 2025, Fig 1b]
To demonstrate the clinical relevance of these PDX zebrafish models for pediatric cancer patients, Dr. Berman knew he needed to compare his results to those obtained using mouse PDX models. He was interested in the work that Dr. Fletcher and colleagues had been doing in Australia because they had detailed data from actual patient outcomes in addition to results in cell lines and mouse PDX models developed directly from these patients. “We had optimized the zebrafish platform in Canada,” commented Dr. Berman. “And we had the patient, cell, and mouse data in Australia,” added Dr. Fletcher.
Comparing Models and Patient Outcomes
As part of their collaboration, Dr. Fletcher and his team sent tumor samples from 10 Australian patients to Dr. Berman’s laboratory in Canada. The patients represented 6 different types of pediatric cancers including Ewing sarcoma, soft-tissue sarcoma, neuroblastoma, osteosarcoma, gastrointestinal stromal tumor, and anaplastic large cell lymphoma.
Each of these 10 patients had been treated in the ZERO program, and the researchers knew which treatments they had received and the outcomes of each treatment. The response to treatment was described by the treatment team according to the Response Evaluation Criteria in Solid Tumors (RECIST) or Positron Emission Tomography Response Criteria in Solid Tumors (PERCIST) criteria.
Dr. Fletcher and his team had successfully created mouse PDX models for 7 of the 10 patients but were unable to make models for 3 of the patients. Using the samples sent by Dr. Fletcher’s team, Dr. Berman and his colleagues developed zebrafish PDX models for each of the 10 Australian patients. They successfully created zebrafish PDX models even for the 3 patients for whom mouse PDX models were unsuccessful.
Dr. Berman and his team tested PDX zebrafish models with the same treatments that Dr. Fletcher had used on the mouse models and compared their results to each other and to what clinicians had observed in the human patients.
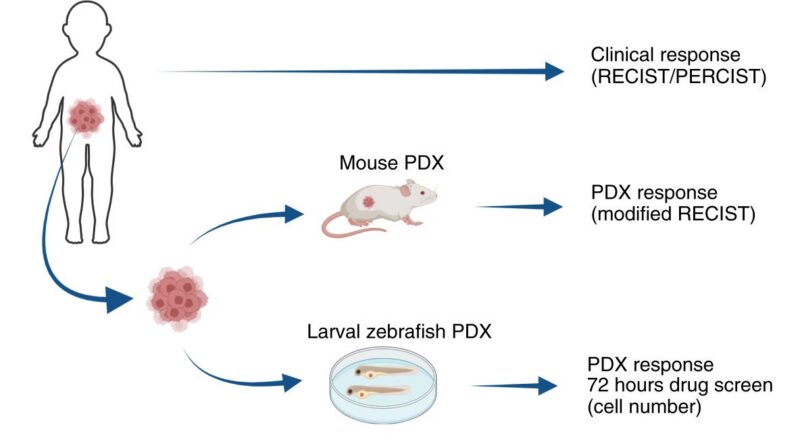
The researchers compared the clinical response of each patient to their treatment based on the Response Evaluation Criteria in Solid Tumors (RECIST) or Positron Emission Tomography Response Criteria in Solid Tumors (PERCIST) criteria to the response of the mouse PDX model based on modified RECIST criteria and the response of the zebrafish PDX model.
[Source: Azzam et al 2025, Fig 1a]
“We were so pleased with how similar our results were,” recalled Dr. Fletcher. The researchers tested a total of 12 treatment options used on the 10 patients. For 11 out of 12 of these options, the zebrafish PDX model results matched the treatment response observed in the human patients. “This is an exciting proof of concept for incorporating PDX zebrafish models into precision medicine workflows for pediatric cancer patients,” commented Dr. Berman.
Future Work
In future work, Dr. Berman and Dr. Fletcher plan to conduct similar analyses for other types of pediatric cancers not included in this study. For example, brain cancers and leukemia were not well represented among the 10 patients, so additional testing is needed in these patient populations. In addition, Dr. Berman continues to refine zebrafish models for understanding pediatric cancers, and Dr. Fletcher is working on improved techniques for conducting precision medicine based on blood samples, which are easier to obtain than tumor samples.
Ultimately, Dr. Berman and Dr. Fletcher envision a world where every child with pediatric cancer has access to all these precision medicine tools, including genetic testing, testing in cell lines, and modeling in both mouse and zebrafish PDX models. “All of these tools have their advantages and disadvantages,” added Dr. Fletcher, “and by working together across models we can get the best picture of how to move forward.” Both researchers emphasized the importance of international collaboration for rare diseases like pediatric cancers. They’ve shown that they can work together, even sending tumor samples back and forth, from opposite sides of the globe. “Science and research really have no borders,” concluded Dr. Berman.
Dr. Jamie Fletcher is Associate Director of Laboratory Research and Principal Scientist at Children’s Cancer Institute in Sydney, Australia, Associate Professor in the Faculty of Medicine at UNSW Sydney, and Co-Scientific Lead of the Preclinical Drug Testing Team at the ZERO Childhood Cancer program. His research focuses on developing precision medicine tools using cell lines and mouse models to improve treatment options for patients with pediatric cancer. When not in the laboratory, Dr. Fletcher enjoys mountain biking, climbing, and spending time with his family.
Dr. Jason Berman is the CEO and Scientific Director of the CHEO Research Institute and VP Research at CHEO in Ottawa, Canada, and is Professor of Pediatrics and Cellular and Molecular Medicine at the University of Ottawa.. He is a practicing pediatric oncologist specializing in pediatric leukemia and a pediatric cancer researcher focused on developing precision medicine tools in zebrafish models. When not leading the CHEO Research Institute, in the laboratory or seeing patients, Dr. Berman enjoys working out on his Peloton, spending time with his family and taking walks with his dog.
- Azzam N, Fletcher JI, Melong N, et al. Modeling High-Risk Pediatric Cancers in Zebrafish to Inform Precision Therapy. Cancer Res Commun. 2025 Jul 1;5(7):1215-1227. doi: 10.1158/2767-9764.CRC-25-0080. https://aacrjournals.org/cancerrescommun/article/5/7/1215/763751/Modeling-High-Risk-Pediatric-Cancers-in-Zebrafish
- Berman Laboratory. https://bermanzebrafishlab.com/wp/
- Precision Oncology for Young People (PROFYLE). https://www.profyle.ca/
- CHEO Research Institute. https://www.cheoresearch.ca/
- Experimental Therapeutics & Molecular Oncology Research Group at the Children’s Cancer Institute. https://www.ccia.org.au/our-research/themes/therapeutic-discovery/experimental-therapeutics-molecular-oncology
- ZERO Childhood Cancer. https://www.zerochildhoodcancer.org.au/home
- American Childhood Cancer Organization. https://www.acco.org/types-of-childhood-cancer/
- National Pediatric Cancer Foundation. https://nationalpcf.org/
- National Cancer Institute. https://www.cancer.gov/types/childhood-cancers


