We do all sorts of things to freshen our breath. Mints, gum, or mouthwash can all do the trick to mask the smell of bad breath or a recent meal. What if some of those smells on your breath could actually tell you something about your health?
In ancient Greece, the physician and philosopher Hippocrates, often called the “Father of Medicine” noted that certain breath odors could indicate disease, like the fruity breath of someone with diabetes1Diabetes: A group of diseases characterized by high blood sugar levels. https://www.niddk.nih.gov/health-information/diabetes/overview/what-is-diabetes. The cause of the sweet smell on the breath of someone with diabetes was eventually identified as acetone, a chemical produced by the breakdown of fats in the body and one of thousands of components that naturally occur in exhaled breath.
Recently, Dr. Huanyu “Larry” Cheng, James L. Henderson, Jr. Memorial Associate Professor of Engineering Science and Mechanics at Penn State University in University Park, Pennsylvania, and colleagues have released a prototype of a sensor that can accurately distinguish between people with and without diabetes based on the quantity of acetone in a sample of exhaled breath. The researchers hope this sensor will contribute to a new era of noninvasive diagnostic testing for diabetes.
Diagnosing Diabetes
According to the American Diabetes Association, over 350,000 children and nearly 40 million adults in the US are living with diabetes. Most people with diabetes in the US have type 2 diabetes2Type 2 diabetes: A type of diabetes where the cells in the body don’t use the hormone insulin properly, leading to high blood sugar levels. https://www.niddk.nih.gov/health-information/diabetes/overview/what-is-diabetes. Type 2 diabetes is a chronic condition that occurs when the body cannot properly absorb glucose, a simple sugar, from the bloodstream, leading to increased levels of glucose in the blood. To learn about additional research on diabetes, see the previous Science Matters stories “Seeing Clearly with Diabetes” and “How to Program Stem Cells.”
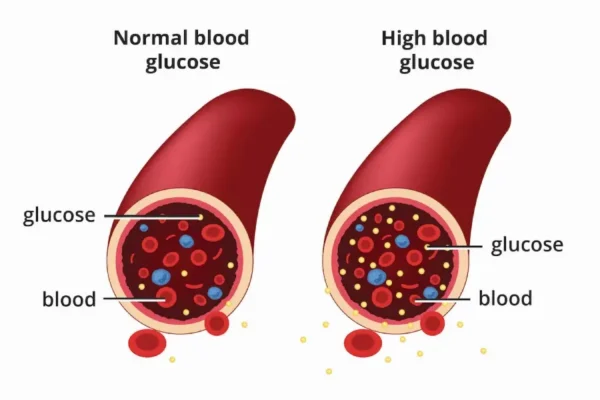
People with diabetes have increased levels of glucose in the blood.
[Source: https://www.niddk.nih.gov/health-information/diabetes/overview/what-is-diabetes]
Over time, high blood glucose levels can lead to a variety of complications including problems with the heart, kidneys, feet, eyes, and skin. These complications can be prevented or delayed through changes in lifestyle and medications.
Preventing complications from diabetes means that you first need to know you have the condition. Health care providers use blood tests taken by either a finger prick or a needle in the vein to diagnose and monitor diabetes. These blood tests determine the level of glucose in the blood in the moment or over the past 2-3 months.
- A glucose test can directly measure the amount of glucose in the blood, usually reported as milligrams of glucose per deciliter (mg/dL). Health care providers have thresholds that they consider to be normal or abnormal depending on your health status. For example, after fasting overnight, the glucose level should be under 100 mg/dL in someone without diabetes.
- A hemoglobin A1C test3Hemoglobin A1C: A test that measures an individual’s average glucose level over the past 2-3 months. https://diabetes.org/about-diabetes/diagnosis can indirectly measure the average glucose levels over the past 2-3 months. Hemoglobin4Hemoglobin: A protein found in red blood cells that transports oxygen from the lungs to the rest of the body. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/hemoglobin is a protein and an important part of red blood cells. Hemoglobin proteins become coated with glucose in the blood over time. The hemoglobin A1C test measures the percentage of hemoglobin proteins that are coated with glucose. In general, an A1C result of 6.5% or greater indicates diabetes.
Glucose monitoring and regular A1C testing are important parts of diabetes management. The results of these tests help guide how a patient and their health care provider make decisions about treatment options. However, the need for bloodwork and medical appointments to understand an individual’s risk for diabetes makes testing burdensome or inconvenient for many people. This is part of the reason that an estimated 25% of adults with diabetes in the US don’t know they have it.
To improve the reach of testing for diabetes, scientists have been investigating additional methods of testing glucose levels that are less invasive. For example, researchers have developed sensors that can detect glucose levels in sweat rather than blood. This test is less invasive than a blood test, but researchers still need to cause patients to sweat, either through exercise, a sauna, or chemicals, to get a sample.
This is where acetone comes in. Acetone was first identified as a marker of diabetes in the mid-1800s, and its utility has been confirmed in both animal models and human studies. For example, studies in rat models of diabetes helped establish the correlation between acetone levels in exhaled breath and blood glucose levels. This was difficult to accomplish solely in human subjects because the specific components of exhaled breath in humans are highly variable.
Despite the scientific understanding of acetone as a marker of diabetes, its use in everyday medical practice was limited by technology. For decades, there were no sensors capable of detecting acetone in exhaled breath and interpreting the results ways that would be relevant for patients. Fortunately, recent advances in technology have now turned the possibility of using acetone as a biomarker for diabetes into reality. This was the goal of Dr. Cheng and colleagues, who specialize in the development of portable devices for a variety of medical applications.
Sensing Acetone in the Airways
Acetone is a naturally-occurring chemical made of hydrogen, oxygen, and carbon. You may be familiar with its distinctive smell, as it’s the main ingredient in nail polish removers and paint removers. Acetone is released into the air during forest fires and volcanic eruptions and is one of many components found in car exhaust and tobacco smoke.
Acetone is also produced naturally in the body as one of the byproducts of breaking down fat for energy. It is released in urine and in breath you exhale. In healthy people, the concentration of acetone in exhaled breath is about 300-900 parts per billion (ppb). In people with diabetes, the amount of acetone in exhaled breath is increased, anywhere from 900-1,800 ppb.
Scientists use several methods to identify and quantify the components of exhaled breath. For example, gas chromatography-mass spectrometry uses a carrier gas5Carrier gas: A non-reactive gas such as helium, hydrogen, or nitrogen used to carry samples for analysis with gas chromatography. https://www.britannica.com/science/carrier-gas to separate the components of a sample and mass spectrometry to identify the components based on their properties.

Gas chromatography-mass spectrometry laboratory equipment.
[Source: https://commons.wikimedia.org/wiki/File:GCMS_closed.jpg]
A related method, proton transfer reaction-mass spectrometry uses hydronium (H30+) to separate the components of a sample instead of a carrier gas.

Proton transfer reaction-mass spectrometry laboratory equipment.
[Source: https://commons.wikimedia.org/wiki/File:PTR-TOF1000_Ultra_Ionicon_mass_spectrometer,_Faculty_of_Chemistry,_Gdansk_University_of_Technology_Poland.jpg]
While effective, both these techniques must be conducted with large, expensive equipment only available in a laboratory setting. “Our goal was to design these technologies to fit on a portable sensor chip while maintaining high accuracy so they can be useful outside of a clinic setting,” explained Dr. Cheng.
Designing A Diabetes Sensor
The most common materials used in sensors that detect components in exhaled breath are metal oxide semiconductors (MOS). MOS combine a metal, such as zinc or titanium, with oxygen. They can be made into different shapes and sizes to serve different purposes.
Previous designs of acetone sensors using MOS were ineffective for clinical use because they needed to be operated at very high temperatures to function. To improve the functionality of MOS-based sensors for acetone, Dr. Cheng and colleagues combined the MOS with graphene, a carbon-based material that is known to function well at room temperature.
The researchers started with a film made of polyimide (PI), a flexible, heat-stable plastic material. They used a carbon dioxide laser to create holes in graphene, called laser-induced graphene (LIG), and placed the LIG on the polymer platform. Zinc oxide (ZnO) solution was added to the LIG using a technique called drip casting. Drip casting uses a liquid dropper to place individual droplets of the solution onto the surface, where they harden into an even sheet. The ZnO layer was covered with a silicon-based material called SBA-15 to protect the sensor from humidity and moisture in exhaled breath.
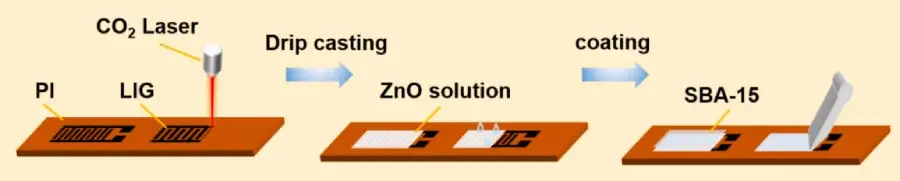
The process of creating a sensor to detect acetone in exhaled breath. The researchers used a carbon dioxide (CO2) laser to create laser-induced graphene (LIG) foam on top of a polyimide (PI) film. They added a zinc oxide (ZnO) solution using a method called drip casting and coated the product with the silicone-based material SBA-15.
[Source: Yang et al 2025, Fig 1a]
The resulting product looked like a sandwich with three layers: a mixture of ZnO and LIG on the bottom, ZnO in the middle, and the protective coating SBA-15 on top.
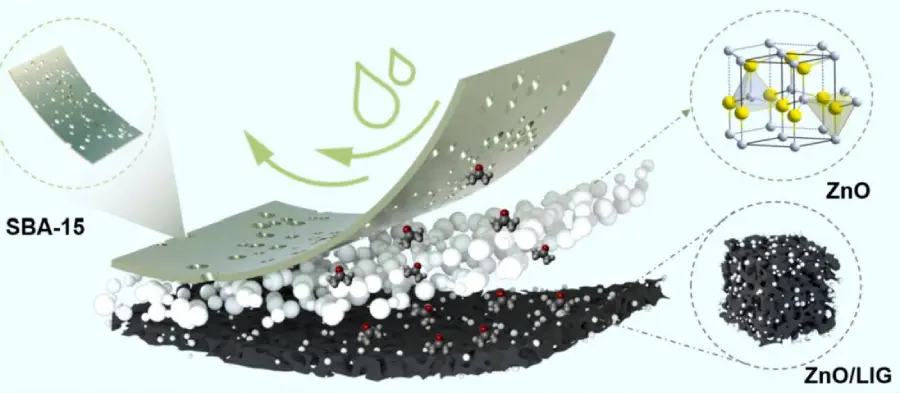
Diagram of the sandwich structure of the acetone sensor. A mixture of zinc oxide (ZnO) and laser-induced graphene (LIG) on the bottom, a layer of ZnO in the middle, and the protective silicone-based coating SBA-15 on top.
[Source: Yang et al 2025, Fig 1b]
Once Dr. Cheng and colleagues had developed the prototype for the acetone sensor, they performed multiple series of experiments to make sure it worked properly in the laboratory. For example, the researchers exposed the sensor to different concentrations of acetone and observed the output for accuracy. They were able to adapt the sensor accordingly. Ultimately, they found the sensor was able to detect the level of acetone down to 4 ppb in less than 30 seconds under laboratory conditions. “We were happy enough with these results that we were ready to test the sensor in real life,” remarked Dr. Cheng.
A Diabetes Sensor Comes to Life
After optimizing the design of the sensor in the laboratory to be as sensitive as possible, Dr. Cheng and colleagues tested the sensor in real life. A total of 71 volunteers participated in this study, including 51 patients with diabetes and 20 healthy volunteers. Each participant provided a sample of their exhaled breath, which was collected in a special gas collection bag for analysis, as well as a blood sample.
The results showed that the sensor prototype could accurately distinguish between patients with and without diabetes based on the amount of acetone in the exhaled breath. This was confirmed by comparing the sensor results with the fasting blood glucose levels of each participant.
In future work, Dr. Cheng plans to continue to improve the capabilities of the sensor. Currently, the sensor works best when exposed to exhaled breath collected in a bag. Ideally, the sensor would be placed in a face mask directly under the nose.
The researchers need to overcome multiple additional challenges to make this possible. For example, inside a face mask, the sensor would face a greater variety of conditions, such as air speed and humidity. “We want to develop this sensor so that it is still accurate in more variable environments,” added Dr. Cheng. To learn about additional research developing face masks, see the previous Science Matters story “Facemasks of the Future.”
Dr. Huanyu “Larry” Cheng is James L. Henderson, Jr. Memorial Associate Professor of Engineering Science and Mechanics at Penn State University in University Park, Pennsylvania. His research focuses on the development of portable device platforms such as sensors. When not in the laboratory, Dr. Cheng enjoys hiking, swimming, and spending time with his family.
