When you go to get a vaccine, it is common practice for the health care provider who will give the shot to ask your preferred location. In babies and young children, vaccines are usually administered in the thigh or buttock, where we have the most muscle. Older children and adults usually receive vaccines in the large muscles of the upper arm. The provider might ask: “Which arm do you want it in?”
Since vaccines can sometimes cause pain or soreness around the injection site due to a localized response from the immune system,1Immune system: A network of organs, tissues, and cells that fight infection and prevent disease. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/immune-system many people prefer their non-dominant arm. Still, this is usually the patient’s choice, since the location of the injection is not thought to impact the effectiveness of the vaccine. However, new research by Australian scientists Dr. Mee Ling Munier, Prof. Anthony Kelleher, and Prof. Vanessa Venturi at the Kirby Institute, UNSW Sydney and Prof. Tri Phan at the Garvan Institute of Medical Research and their colleagues shows that the location of the injection site might be more important than previously thought. They hope this research will lead to a deeper understanding about how vaccines protect us from diseases as well as inform future public health practices in the case of another pandemic.2Pandemic: Outbreak of disease that spreads over a wide geographical area, such as multiple countries or continents. https://www.merriam-webster.com/dictionary/pandemic
The Immune System: Our Body’s Best Defense
There are dangers to our physical well-being all around us—infection-causing microbes3Microbes: Microscopic organisms such as bacteria and viruses. https://www.ncbi.nlm.nih.gov/books/NBK279387/ in the air and water, diseases transmitted by insects, rodents, and other animals, and possible injuries around every corner, just to name a few. Most of the time, we don’t think about these dangers because our bodies have a remarkable capacity to protect us from disease and heal when we do get sick or injured.
Our best defense against these dangers is the immune system4Immune system: A network of cells, tissues, and organs that prevent and fight infection and illness. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/immune-system, a network of organs, tissues, and cells that work together to prevent us from getting sick and help us get better when we do. Organs of the immune system include the skin, lymph nodes5Lymph nodes: Small structures throughout the body that are part of the immune system; they filter lymph fluid and contain white blood cells that fight disease. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/lymph-node , tonsils6Tonsils: Lymph node-like masses on either side of the throat that are part of the immune system. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/tonsil , spleen7Spleen: An organ of the immune system that filters blood and produces white blood cells. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/spleen, and bone marrow8Bone marrow: Spongy tissue at the center of most bones that produces stem cells that can become blood, fat, and bone cells. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/bone-marrow. Anything that triggers an immune response is called an antigen9Antigen: Any substance that produces a response from the immune system, such as bacteria, viruses, toxins, and other chemicals. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/antigen. Both microbes that cause disease and some vaccines that prevent disease contain proteins that are considered antigens because they interact with the immune system.
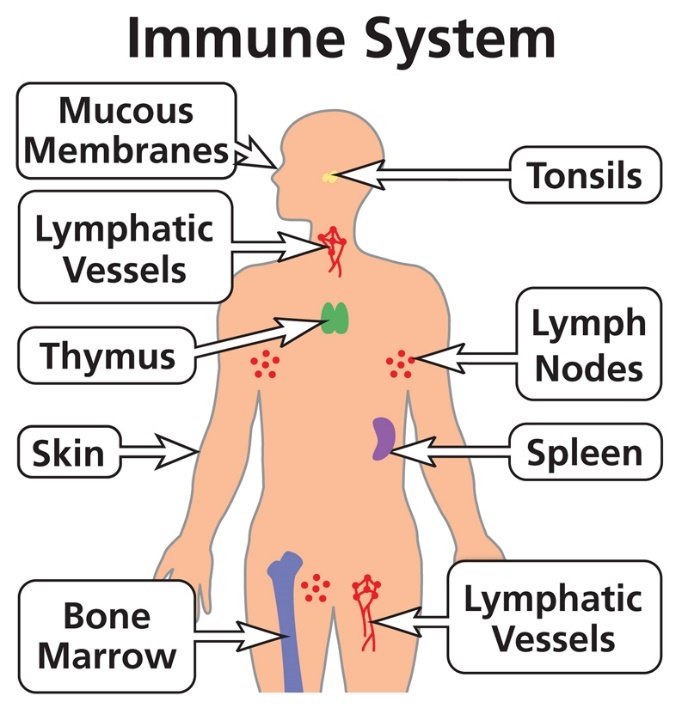
Figure 1.
Organs of the immune system.
[Source: https://clinicalinfo.hiv.gov/en/glossary/immune-system]
Broadly speaking, the immune system is divided into two parts: the innate immune system and the adaptive immune system10Adaptive immune system: A specialized part of the immune system that remembers potentially harmful substances it has come in contact with to prevent future disease. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/adaptive-immunity.
- The innate immune system is the body’s first line of defense against microbes that can cause disease. It is considered non-specific because it reacts similarly to any type of antigen.
- The adaptive immune system is considered specific because it is activated only by specific antigens.
The innate immune system includes skin, mucus, and stomach acid:
- The skin acts as a barrier to microbes entering the body.
- Mucus in the nose and mouth traps small particles.
- Stomach acid kills microbes that enter our bodies through food or drinks that we consume.
The innate immune system also involves different cell types that immediately attack any antigens that make it through this first line of defense. For example, macrophages11Macrophage: A type of white blood cell that ingests microorganisms and stimulates other parts of the immune system. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/macrophage are one of the first immune cells to encounter an antigen. They envelop the antigen and break it down into fragments.
The adaptive immune system includes immune cells that have learned to recognize certain antigens over time, allowing them to react more quickly a second time. This is why you typically don’t get the exact same cold twice—the adaptive immune system learns to recognize the virus and can more easily fight against it next time. However, because this reaction is specific to particular antigens, the body is still vulnerable to a different virus—or a similar virus with enough changes that the adaptive immune system no longer recognizes it.
A variety of immune cell types are involved in the adaptive immune system, including:
- T cells12T cells: A type of white blood cell that destroys infected cells and signals other immune cells, such as B cells, to fight infection. https://my.clevelandclinic.org/health/body/24630-t-cells recognize antigen fragments and activate B cells13B cells: A type of white blood cell that produces antibodies to fight infection and remembers antigens to attack in the future. https://my.clevelandclinic.org/health/body/24669-b-cells, another type of immune cell.
- B cells are activated by T cells to become either plasma cells14Plasma cells: A type of white blood cell derived from B cells that produces large amounts of specific antibodies. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/plasma-cell or memory B cells15Memory B cells: A type of B cell that remembers antigens so that the immune system can work more quickly in the future. https://my.clevelandclinic.org/health/body/24669-b-cells.
- Naïve16Naïve cells: White blood cells that circulate in the body but have not yet been activated by an antigen. https://www.akadeum.com/blog/what-are-naive-cells/?cn-reloaded=1 B cells have not yet encountered an antigen. Plasma cells are B cells that produce antibodies17Antibodies: Proteins produced by B cells of the immune system to recognize foreign substances and neutralize them. https://www.genome.gov/genetics-glossary/Antibody, proteins that bind to specific antigens and neutralize them. Memory B cells “remember” the antigen they are activated to respond to, so they can respond more quickly the next time.
Different types of immune cells of the innate and adaptive immune systems. Macrophages are part of the innate immune system; T cells, B cells, and the antibodies produced by B cells are part of the adaptive immune system.
[Source: https://commons.wikimedia.org/wiki/File:Innate_and_Adaptive_Immunity.png]
B cells are produced in the bone marrow, the spongy tissue at the center of our bones. Once B cells are activated to become either plasma cells or memory B cells, they may travel to other parts of the immune system, namely the spleen and the lymph nodes, or return to the bone marrow. There are hundreds of lymph nodes in the body including in the underarms, neck, abdomen, and groin.
When B cells are exposed to an antigen, such as an infection or vaccine, they form temporary structures called germinal centers in the lymph nodes and other organs of the immune system. Germinal centers act like “schools,” where B cells can go to learn about the antigen. With this information, plasma cells develop antibodies against the antigen, and memory B cells remember it so they can recognize it in the future.
Vaccines Boost the Immune System
The idea behind vaccines is to expose the body to certain antigens just enough so that the adaptive immune system develops the ability to recognize it, but not enough to make us sick. If the vaccine is effective, the next time the body is exposed to the same antigen, it can quickly recognize and fight it, so we don’t get sick. For some diseases, a series of vaccines given weeks, months, or years apart may be needed to provide full protection.
Recent research in animal models has changed scientific understanding of how memory B cells interact with vaccines. Prof. Tri Phan, Program Director of Precision Immunology at the Garvan Institute of Medical Research, is a physician-scientist whose research focuses on the role of B cells in the immune system. His work has shown that B cells only form germinal centers in the lymph node closest to the vaccination site (called the draining lymph node in this research). It was previously thought that B cells would take what they learned in germinal centers and circulate throughout the body to look out for specific microbes. However, Prof. Phan, along with former PhD student Rama Dhenni, and colleagues showed that this isn’t always the case. Although some memory B cells do circulate, others stay behind in the draining lymph node.
Prof. Phan and colleagues also uncovered a distinct role for macrophages in the draining lymph node. Although macrophages are usually known as large immune cells that ingest antigens, Prof. Phan found that macrophages in germinal centers have a different, specialized role. His research shows that vaccination primes these macrophages to interact differently with memory B cells. Rather than ingesting antigens, these macrophages are sensitive to the virus they learned about in the germinal center. When they come in contact with the virus—either through infection or a booster18Booster: An additional vaccine given after the person has had the initial vaccine in order to “boost” the ability of the immune system to recognize and fight a particular antigen. https://kidshealth.org/en/parents/booster-shots.html vaccine—they alert memory B cells to return to the germinal center.
Figure 3.
A video of cell movement and behavior inside the draining lymph node of a mouse (blue). The memory B cells (red) stay around the same area and interact with macrophages (white).
[Source: Dr. Rama Dhenni]
In mice, Prof. Phan’s research suggested that receiving a booster vaccine on the same side might be beneficial to re-activating the immune system and boosting the effectiveness of the vaccine. The two-series vaccinations developed against SARS-CoV-2, the virus that causes COVID-19, provided an opportunity to test whether this might also be true in humans.
The Human Element
Dr. Mee Ling Munier is a Senior Lecturer and Group Leader of the Vaccine Immunogenomics Group within the Immunovirology and Pathogenesis Program at the Kirby Institute in Sydney, Australia. Her research focuses on the immune response to infection and vaccination, and she works closely with Prof. Phan and many other collaborators.
Dr. Munier, PhD student Alexandra Carey Hoppé, and colleagues recruited 30 healthy volunteers who had never been infected with SARS-CoV-2 and who had not yet received the vaccine. The researchers confirmed that participants had never been infected with SARS-CoV-2 by testing their blood for antibodies. The volunteers received the series of two Pfizer vaccinations (BNT162b2) three weeks apart. Twenty volunteers received the second dose in the same arm as the first, and 10 volunteers received the second dose in the opposite arm. Bloodwork was collected before the second vaccination, 4 weeks after the second vaccination, and 4-6 months after the second vaccination. Eighteen participants also agreed to undergo a fine needle biopsy19Fine needle biopsy: A specialized technique that uses a needle inserted into the lymph node to remove a sample of the lymph fluid it contains. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/fine-needle-aspiration-biopsy 5-7 days after the second vaccination.
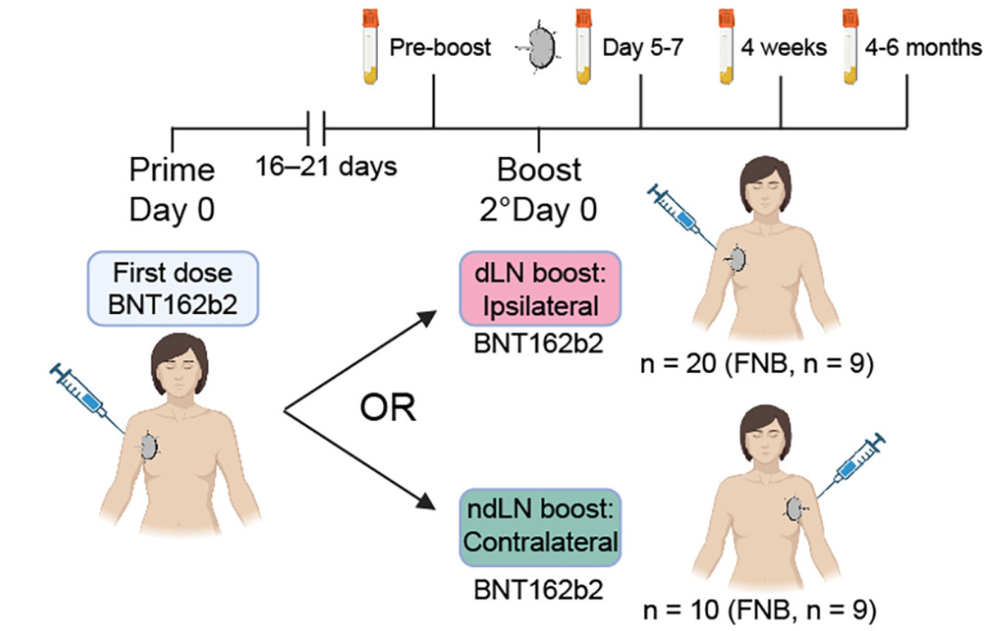
Flow of the research project. Thirty participants were given two doses of the SARS-CoV-2 Pfizer vaccine (BNT162b2). Twenty participants received the second dose (boost) in the same side (ipsilateral or draining lymph node (dLN)) and 10 received the second dose on the opposite side (contralateral or non-draining lymph node (ndLN)). All participants had bloodwork drawn before the boost and 5-7 days, 4 weeks, and 4-6 months after. Nine participants in each group also had fine needle biopsy (FNB) performed 5-7 days after the boost.
[Source: Dhenni et al. 2025, Figure 7a]
Fine needle biopsy is a specialized technique that uses a needle inserted into the lymph node to remove a sample of cells20Lymph: A clear fluid carrying white blood cells and other substances that is circulated throughout the lymphatic system by lymph vessels and filtered in lymph nodes. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/lymph.
Members of Dr. Munier’s laboratory perform a fine needle biopsy of the lymph node.
[Source: Dr. Munier]
The researchers analyzed the lymph node samples using fluorescent proteins to identify the presence of different immune cells including naïve B cells, germinal center cells, B cells, plasma cells, and memory B cells. They compared the relative proportions of these cells in the boosted draining and non-draining lymph nodes and immediately saw important differences. In the lymph node samples of draining lymph nodes, there were many more germinal center B cells and plasma cells than in the non-draining lymph nodes, indicating a heightened response to the booster vaccination.
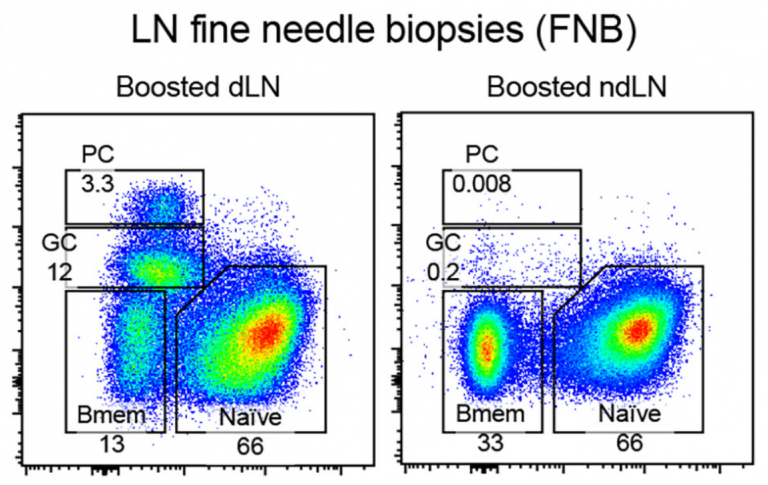
Relative proportions of B cells in the tissue sample of a fine needle biopsy performed on a boosted draining lymph node (dLN) and non-draining lymph node (ndLN). There are many more plasma cells (PCs) and germinal cells (GCs) in the boosted dLN compared to the boosted ndLN.
[Source: Dhenni et al. 2025, Figure 7b]
Differences in the amount of antibodies produced by B cells that respond to the vaccine were also observed in bloodwork results taken at the same time as the fine needle biopsies. However, these differences became less pronounced over time. There was no statistical difference between the two groups after 4 weeks and 4-6 months.

Dr. Munier in the laboratory with graduate students Alexandra Carey Hoppé and Mollie Boyd.
[Source: Dr. Munier]
Overall, these results show differences in vaccine effectiveness in the early time period just after boosting that indicate it may be most effective to receive vaccines in the same series in the same location. Although these differences do decrease over time, extra immunity might be especially important during a future epidemic21Epidemic: An outbreak of disease that spreads quickly and affects many people at the same time. https://www.merriam-webster.com/dictionary/epidemic or pandemic. “What’s so great about this research project is that, because we have the data in animal models, we know why we are seeing these differences,” added Dr. Munier. In future work, Dr. Munier plans to conduct further analysis and perform additional experiments to dive deeper into understanding the behavior of different immune cells that respond to vaccines.
Dr. Mee Ling Munier is a Senior Lecturer and Group Leader of the Vaccine Immunogenomics Group within the Immunovirology and Pathogenesis Program at the Kirby Institute in Sydney, Australia. Her research focuses on the immune response to infection and vaccination. When not in the laboratory, Dr. Munier enjoys spending time with her family and pets.
Professor Tri Phan is a physician-scientist and Program Director of Precision Immunology at the Garvan Institute of Medical Research in Sydney, Australia. His research is focused on B cell macrophage interactions in immunity, autoimmunity and cancer. When not in the lab he enjoys the cryptic crossword and walking aimlessly.
Associate Professor Vanessa Venturi is a computational biologist with the Immunovirology and Pathogenesis Program at the Kirby Institute, UNSW Sydney, Australia. She uses her mathematical, computational and problem-solving skills to analyze and interpret complex data with the aim of better understanding how the immune system responds to infection and vaccination. When not working, Vanessa enjoys reading and doing puzzles and word and logic games with her family.
Professor Anthony Kelleher is a clinician-scientist, immunologist and Director of the Kirby Institute, UNSW Sydney, Australia. His research has impacted HIV treatment and prevention policies nationally and globally. When not working, Tony enjoys bushwalking and body surfing.
- Dhenni R, Hoppé AC, Reynaldi A, et al. Macrophages direct location-dependent recall of B cell memory to vaccination. Cell. 188(13):3477-3496 (2025). https://www.cell.com/cell/fulltext/S0092-8674(25)00407-6
- Dr. Munier’s Laboratory. https://www.kirby.unsw.edu.au/our-people/mee-ling-munier
- Prof. Phan’s Laboratory. https://www.garvan.org.au/research/labs-groups/phan-lab
- This Could Change the Arm You Get Vaccinated. Health Report. https://www.abc.net.au/listen/programs/healthreport/vaccination-site-immune-system/105234612
- Vaccines Explained. World Health Organization. https://www.who.int/teams/immunization-vaccines-and-biologicals/diseases/explainers
- Immunizations. American Academy of Pediatrics. https://www.healthychildren.org/English/safety-prevention/immunizations/Pages/default.aspx
- Written by Rebecca Kranz with Andrea Gwosdow, PhD at http://www.gwosdow.com